3D Bioprinting:
Growing a Child’s Own Ear in the Lab
What if a child’s own cartilage cells could be harvested, multiplied in the laboratory, and printed into a precise three-dimensional ear framework — eliminating the need for rib harvest entirely? Dr. Bonilla served as a clinical investigator in this clinical trial — the world’s first FDA-authorized human clinical trial of 3D-bioprinted ear cartilage for microtia reconstruction. While that specific trial has concluded, similar research continues to advance in the field and Dr. Bonilla remains actively engaged in following its development. In plain terms: this technology uses a patient’s own cells to grow cartilage, rather than relying on synthetic materials — a fundamental difference from any implant-based approach.
What Is 3D Bioprinting of Ear Cartilage?

Three-dimensional bioprinting is an additive manufacturing process — like an industrial 3D printer, but instead of plastic or metal, it deposits living biological material. In the context of microtia reconstruction, the process involves growing a patient’s own cartilage cells in a laboratory, mixing them into a printable biological “ink,” and using a precision printer to build a three-dimensional ear framework — layer by layer — in the exact shape and dimensions needed for that specific child.
The critical distinction from all prior synthetic alternatives is one word: autologous. The cells used to build the ear framework are the patient’s own. Not a donor’s. Not manufactured polymer. The child’s own cartilage genetics, the child’s own biological signature — printed into the shape of an ear.
If successful at clinical scale, this technology would address the one genuine limitation of natural rib cartilage reconstruction — the need to harvest cartilage from the child’s chest. It would preserve everything that makes living autologous tissue superior (integration, growth, no rejection, natural feel) while eliminating the chest incision and the wait for sufficient rib cartilage maturity.
This is why the research community’s focus is here — not on better synthetic implants, not on prosthetics — but on growing living tissue in the laboratory. The scientific consensus on what the ideal ear framework is made of has never changed: the patient’s own living cartilage. Bioprinting is the attempt to produce that result without the harvest.
The bioink: In this bioprinting approach, chondrocytes (cartilage cells) harvested from a small tissue sample are multiplied in culture and suspended in a hydrogel matrix that provides structural support during printing. The resulting “bioink” maintains cell viability through the printing process and allows the framework to mature into genuine cartilage tissue as it integrates after implantation.
How 3D Bioprinting of Ear Cartilage Works — Step by Step
The technology involves four stages that transform a small tissue sample from the patient into a precision-grown three-dimensional ear framework.
Why this approach is biologically correct: Unlike synthetic implants, a bioprinted ear framework contains living cells from the start. The goal of maturation after implantation is not to create something new — it is to allow a living structure that already exists to complete its biological integration. This is fundamentally different from asking synthetic material to behave like tissue. It is providing tissue and allowing it to become what tissue naturally does.
The Clinical Trial: An Early FDA-Authorized Effort in 3D-Bioprinted Ear Cartilage Reconstruction
The clinical trial represented a landmark moment in the history of microtia treatment — and in the broader field of regenerative medicine. For the first time, a regulatory authority authorized the testing of a living, three-dimensionally bioprinted biological structure as a medical implant in human patients.
The FDA’s Breakthrough Device Designation acknowledges that this technology addresses a serious condition for which no existing alternative provides the combination of biological integration, growth potential, and precision shaping that bioprinting theoretically offers.
Early results from the trial — published in peer-reviewed literature — have shown promising preliminary findings with respect to safety and early structural maintenance. The full assessment of long-term outcomes was ongoing at the time of publication.
Why 3D Bioprinting Could Change Microtia Treatment
If this technology achieves clinical validation, the implications go beyond eliminating a chest scar. They touch every dimension of what makes microtia reconstruction challenging.
From Laboratory Research to Human Trial — The Development Journey
Tissue engineering for ear reconstruction has been an active research goal since the 1990s. Here is how the science arrived at its current stage.
Dr. Bonilla’s Role in the Clinical Trial — and What It Means for His Patients
Dr. Bonilla was invited to participate in this trial because of the depth and exclusivity of his clinical focus — 30 years of practice dedicated entirely to pediatric microtia, with thousands of rib cartilage reconstructions and a pattern recognition in this specific anatomy that only comes from that kind of singular specialization.
That expertise makes him uniquely valuable as a clinical investigator in a trial where understanding exactly what a successful outcome looks like — in this specific anatomy, in this specific age group, across this specific procedure — requires the kind of pattern recognition that only comes from exclusive specialization at high volume.
For families who were patients at Dr. Bonilla’s practice during the trial, his involvement represented direct access to the most advanced emerging work in microtia reconstruction — from a surgeon who both performs the current gold standard and actively contributed to its development.
What We Know, What We Don’t Know, and What This Means for Families Today
This Is Emerging Technology — Not Yet Standard of Care
3D bioprinting of ear cartilage was studied in a clinical trial and is not yet an available treatment. The clinical trial was a Phase 1/2a study — designed to establish safety and generate initial efficacy data. Even with the FDA’s Breakthrough Device Designation and genuinely promising early results, the path from first-in-human study to standard-of-care clinical practice involves rigorous evidence accumulation and regulatory review that cannot be rushed or shortcut.
What we know: The technology works at the biological level — bioprinted autologous cartilage can be produced, implanted, and maintains early structural integrity. Early safety data has been encouraging. The first-in-human results have been published and reviewed. The regulatory pathway is established and active.
What we don’t yet know: Long-term structural permanence — does the bioprinted framework maintain its shape and integration at 5, 10, 20 years the way rib cartilage does? Does it grow proportionally with the child’s face? What are the complication rates across a larger patient population? These are exactly the questions the trial was designed to answer.
What this means for families making decisions today: If your child is ready for reconstruction now, rib cartilage reconstruction with natural tissue remains the gold standard — 50+ years of outcomes data, proven growth, proven permanence, no uncertainty. Dr. Bonilla performs this with a depth of experience that is unmatched anywhere in the world. That is the right choice for most families today.
If your family has the ability to wait, the field continues to advance through ongoing research in regenerative medicine. But Dr. Bonilla will not encourage a family to delay a clinically indicated reconstruction on the hope of accessing a technology that has not yet completed its evidence base. The standard of care today is rib cartilage. The aspiration for tomorrow is bioprinting. Both are worth understanding.
The Gold Standard Is Still Here — and Still Extraordinary
It is easy, reading about bioprinting, to treat rib cartilage reconstruction as something to be superseded. That framing is premature. Natural rib cartilage reconstruction — in experienced hands — produces permanent, living, biological ears that grow with children for life, with no foreign material, no rejection risk, no replacement timeline, and 50+ years of documented outcomes. It is a genuinely remarkable procedure.
Bioprinting’s goal is to produce the same result without the one limitation of rib cartilage: the chest harvest. If it achieves that, it will represent an incremental improvement on something already very good. The promise of something better in the future does not diminish what is exceptional today.
Dr. Bonilla will continue performing rib cartilage reconstruction for every patient for whom it is the right choice today — while simultaneously advancing the science that may make tomorrow’s surgery even better.
What Families Ask About 3D Bioprinting and This Clinical Trial
The clinical trial has concluded its enrollment phase. Eligibility was determined by specific criteria including age, microtia grade, prior surgical history, and other clinical factors. As the trial was in an early phase, enrollment was limited to patients who met specific parameters designed to optimize safety and data quality.
While that specific trial is no longer enrolling, Dr. Bonilla can discuss your child’s situation at consultation and provide an honest assessment of whether any emerging research opportunities may be relevant — or whether rib cartilage reconstruction is the appropriate path forward now.
This is the question Dr. Bonilla is asked most frequently about the trial — and he has a direct answer: for most families, the answer is no.
Rib cartilage reconstruction is ready. It has 50+ years of outcome data, it produces living, permanent ears, and in Dr. Bonilla’s hands the results are outstanding. Waiting for a technology that has not yet completed its evidence base — with no guaranteed timeline for approval — is not a decision he would encourage.
The exceptions are families whose children are not yet at the age for rib cartilage reconstruction, or families who have a specific reason to wait and wish to explore trial eligibility. For those families, Dr. Bonilla can assess the situation individually.
Honest answer: impossible to predict with confidence. Translational medicine timelines depend on trial completion, regulatory review, manufacturing scale-up, and real-world implementation — all of which involve variables that are genuinely difficult to forecast.
What can be said: the technology was taken through a human clinical trial, not merely theoretical research. The FDA recognized its potential with Breakthrough Designation. Early results were encouraging. The direction of progress was meaningful — but “available for all patients” is not something anyone can responsibly put a specific year on, and similar research continues to advance.
If successful, bioprinting would replace the need for rib cartilage harvest entirely as well as the need for Medpor/Su-Por entirely for patients who receive it.
It is worth noting that even in the bioprinting approach, a small tissue sample is still needed from the patient to provide the initial cells. This is a much smaller and simpler harvest than rib cartilage — but it is not entirely harvest-free in the strict sense. The difference is the scale and site of the tissue collection: a tiny sample vs. a cartilage block from the chest ribs.
The general use of the term “3D printed” is slightly misleading. These 3D-printed synthetic plastic ears are actually molded from a CT scan image or a scan performed in the office — they are custom-shaped polyethylene or other plastic. This is still a synthetic implant with all the properties of synthetic implants: inert, does not grow, foreign body risks, extrusion risk. It is not the same at all as a 3D-bioprinted ear made from the patient’s own living cells — such as the one Dr. Bonilla implanted.
This approach and 3D bioprinting are categorically different from synthetic implants. The framework is not synthetic material — it is living cells suspended in a biological matrix, printed into shape. The goal is a living, biological ear framework made from the patient’s own cells. The “3D” part is the manufacturing method, not the material. The material is the patient’s own living cartilage — grown and shaped outside the body, then implanted.
Bioprinted cartilage research has implications across reconstructive surgery broadly — not only microtia. For cartilage repair in trauma, joint surgery, and other applications, the ability to grow patient-specific cartilage would be transformative.
Within microtia specifically, Dr. Bonilla’s practice is exclusively pediatric — treating patients from newborn through age 17. The research aims to improve on the already-excellent natural rib cartilage technique for his pediatric patients — particularly in cases where rib cartilage availability is limited. Whether future trials expand beyond pediatric populations is a question for the research teams conducting those trials.
When the results of the 3D bioprinting clinical trial were published, coverage appeared in more than 100 media outlets across six continents.
Dr. Bonilla served as the principal clinical investigator. The following outlets covered the story independently — in print, broadcast, and digital media.
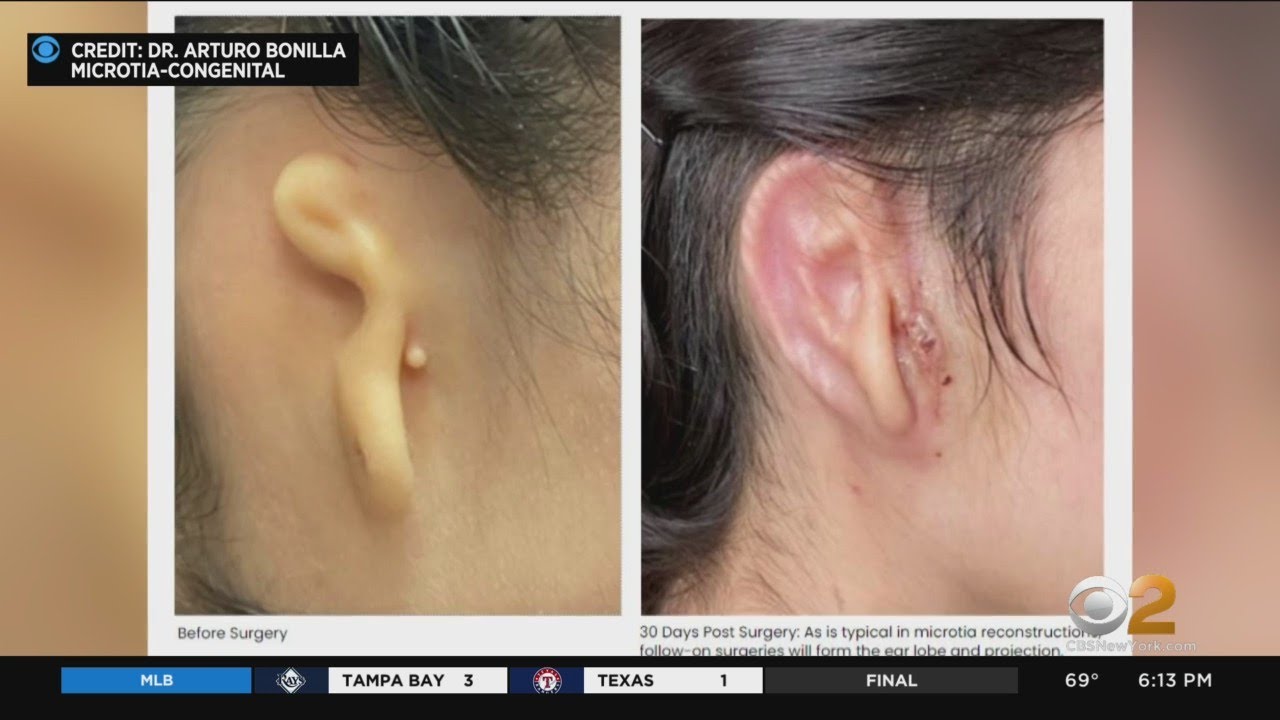
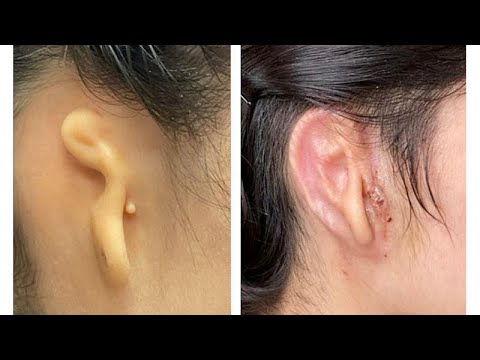

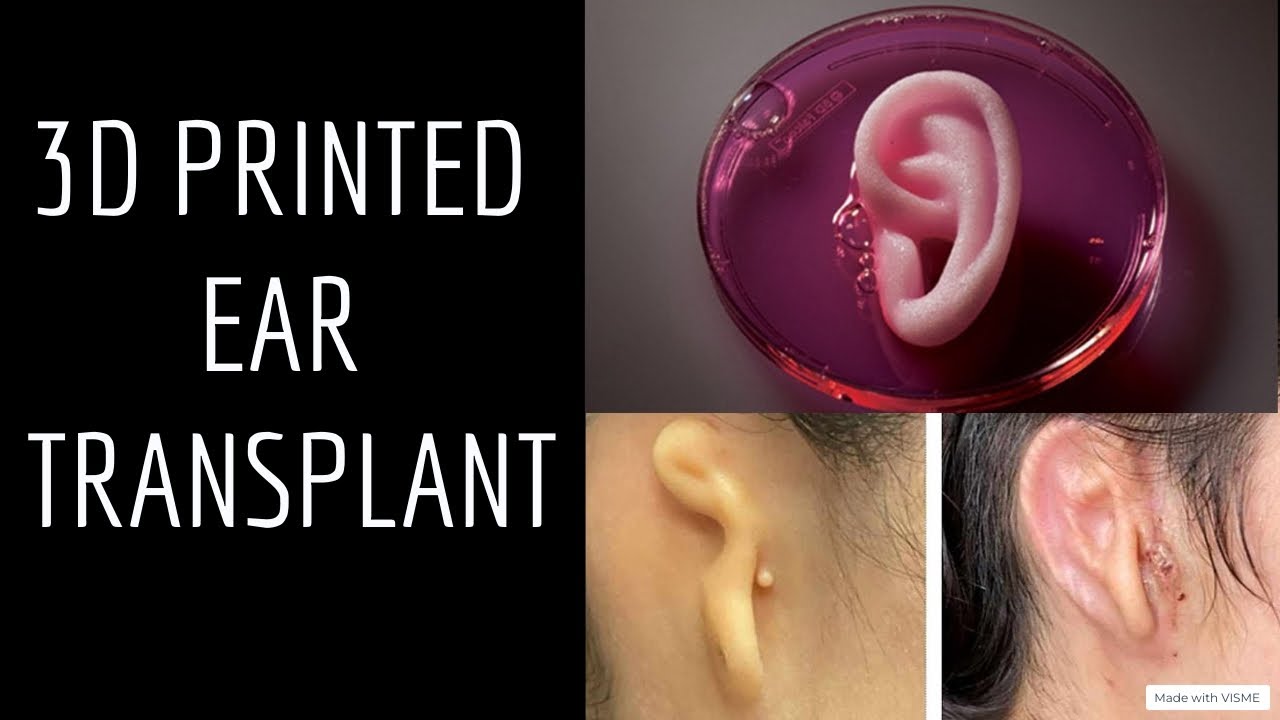

"As a physician who has treated thousands of children with microtia from across the country and around the world, I am inspired by what this technology may mean for microtia patients and their families."
"I've actually been waiting for this my whole career."
- Doctors Transplant Ear of Living Tissue — The New York Times
- Woman gets 3D printed ear transplant made of her own cells — CBS News
- Woman's ear rebuilt with 3D-printed living tissue implant — The Guardian
- 'Groundbreaking' Surgery Gives Woman a 3-D Printed Ear Made of Human Cells — People Magazine
- Doctors perform first successful transplant of 3D-printed ear made from living cells — NBC News
- Surgeons Transplant 3D-Printed Ear Made From Patient's Own Cells — Smithsonian Magazine
- A surgically implanted, 3D-printed ear marks a medical advance — Yahoo News
- Woman born with rare deformity gets 3D-printed ear — Daily Mail
- First 3D printed ear made with human cells transplanted — Axios
- Woman becomes first to get new 3D printed ear made from her own cells — Sky News
- The most powerful health innovations of 2022 — Popular Science
- Cornellian-founded company implants 3D-bioprinted ear — Cornell Chronicle
- In a World's First, Scientists Transplant 3D-Printed 'Living Ear' Onto Patient — CNET
- Lab-Grown Ear Made From Patient's Own Cells And Successfully Implanted — IFLScience
- World-first implant of 3D-bioprinted ear made from patient's own cells — New Atlas
- A Patient Has Successfully Received a 3D Printed Ear Transplant — 3D Natives
- A woman receives the first-ever successful transplant of a living, 3D-printed ear — ZME Science
- New surgery implants living, 3D-printed body parts — Freethink
- Doctors Transplant 3D Printed Ear Made of Living Human Flesh — Futurism
- Doctors successfully transplant a 3D-printed ear made of living cells — Interesting Engineering
- 3DBio, Microtia-Congenital Ear Deformity Institute implant 3D-printed ear — Medical Device Network
- 3DBio Therapeutics implants 3D-printed ear in first-in-human trial — NS Medical Devices
- Woman receives first-of-its-kind 3D printed ear implant — Advisory Board
- Human ear reconstructed with 3D-bioprinted implant — Lab + Life Scientist
- 3DBio prints a path to the clinic with living ear implant — Fierce Biotech
- Doctors Successfully Transplant Human Ear Made of 3D-Printed Living Tissue — Medical Design & Development
- Researchers conduct human ear reconstruction using 3D-Bioprinted Living Tissue Implant — Plastic Surgery Practice
- Cirujanos logran implantar oído impreso en 3D hecho de células vivas — El Sol de México
- Miracle! In a world's first, this woman got an ear transplant with 3D Printing process! — Hindustan Times
- US woman gets 3D-printed ear made from her own cells — South China Morning Post
- Doctors transplant 3D-printed ear made of human cells — The Independent
At the Forefront of Microtia Innovation
Whether your family is interested in emerging research or pursuing rib cartilage reconstruction today, Dr. Bonilla can discuss your child’s specific situation and options at consultation.
To get started, submit your information through our contact form and Dr. Bonilla’s team will be in touch to schedule a virtual consultation.
